Medical Grade LiFePO4 Battery Solutions
Reliable Power for Medical Devices, Patient Monitors, Infusion Pumps & Healthcare Equipment
Custom medical battery packs and OEM medical device battery manufacturer — supplying lithium batteries for medical applications including portable medical devices, battery-powered medical carts, medical computer carts with battery, medical UPS battery backup, and battery backup for home medical equipment worldwide.

Why Choose Our Medical Batteries?
From battery-powered medical devices and portable patient monitors to battery-operated medical carts and medical refrigerator battery backup, our LiFePO4 medical battery packs are engineered to the exacting safety and reliability standards demanded by healthcare environments — with full IEC 60601-1 compliance and medical-grade quality assurance at every stage of production.
Medical Grade Certified — IEC 60601-1
Our medical batteries comply with IEC 60601-1 for medical electrical equipment. Every medical battery pack undergoes rigorous testing including biocompatibility assessment, EMC testing, and safety validation — meeting the standards required by medical device battery manufacturers and custom battery pack manufacturers for medical equipment worldwide.
Redundant Dual-Layer Protection
Smart medical batteries with dual protection circuits ensure maximum patient safety. Our medical-grade BMS provides real-time monitoring of voltage, current, temperature, and state of health — essential for battery pack systems for medical devices operating in critical care environments.
Reliable Runtime for Battery-Powered Medical Devices
Extended medical battery life with consistent power delivery throughout discharge. Designed to meet the runtime demands of battery-operated medical devices — from portable ventilators and infusion pumps to battery-powered medical monitors and mobile medical carts — with 4–12 hours of typical operation per charge.
Clean Room Assembly & Medical-Grade Packaging
Clean room assembly, sterile packaging, and custom medical battery labels available for OEM integration. Full traceability and lot control in medical battery manufacturing ensures regulatory compliance and supports medical device battery requirements across global healthcare markets.
Compatible Medical Device Categories & OEM Applications
Medical Battery Configurations
Standard and custom LiFePO4 lithium battery packs for medical devices — from small battery-operated medical devices and portable medical battery packs to high-capacity battery backup systems for emergency medical equipment, medical carts, and battery-powered mobile medical carts

Portable Medical Devices
3.7V – 12V
2–10Ah | Battery-operated medical suction units, medical thermometer battery, diagnostic handhelds, medical optical devices

Patient Monitors
12V – 24V
10–30Ah | Vital signs monitors, ECG machines, battery-powered medical monitors, GE Healthcare Marquette compatible medical battery

Infusion Pumps & Medical Carts
12V – 24V
20–50Ah | Infusion pumps, battery-powered medical computer carts, battery-powered mobile medical carts, hot-swappable medical battery systems

Ventilators & Mobility Aids
24V – 48V
50–100Ah | Portable ventilators, medical scooter batteries, drive medical scooter battery, wheelchairs, medical transport battery

Custom OEM Solutions
Any Voltage
Custom Capacity | Custom battery packs for medical devices, custom medical battery manufacturers, battery pack suppliers for medical equipment
Custom Medical Battery Design — OEM & ODM
We are an experienced medical device battery manufacturer offering full OEM and ODM battery services. Whether you need custom battery packs for medical devices, battery pack systems for medical devices, or custom battery systems for medical equipment, our engineering team works directly with your medical device specifications to deliver compliant, reliable, and manufacturable solutions.
Our custom medical batteries serve applications from portable medical battery packs and battery powered medical computer systems to large-scale medical UPS battery backup and battery backup systems for emergency medical equipment handling.
- IEC 60601-1 and IEC 62133 compliance support from design through production
- Redundant dual-layer protection circuits for life-critical applications
- Clean room assembly and sterile packaging for medical OEM integration
- Custom form factors — cylindrical, prismatic, pouch, and split-pack configurations
- Medical-grade BMS with SMBus, I²C, RS485, or CAN communication
- Full lot traceability and regulatory documentation for medical battery manufacturing
- Hot-swappable medical battery system designs for zero-downtime operation

Why LiFePO4 for Medical Devices?
LiFePO4 (lithium iron phosphate) is the safest, most reliable lithium battery chemistry for medical applications — used in battery systems for medical devices from portable patient monitors and infusion pumps to battery-powered medical carts and emergency medical equipment
Safe Chemistry — No Thermal Runaway
The only responsible choice for battery-powered medical devices operating near patients — zero risk of thermal runaway in clinical environments
2000+ Cycle Life
Long medical battery lifespan reduces total cost of ownership for medical equipment batteries in hospitals, clinics, and home medical settings
Stable, Flat Discharge Voltage
Consistent power delivery throughout the discharge cycle — critical for precision medical battery-powered instruments and diagnostic devices
High Energy Density
Compact lithium medical battery packs with maximum capacity — enabling longer runtime for portable medical battery packs and battery-operated medical devices
Wide Temperature Stability
Reliable operation from -20°C to 60°C — suitable for battery-powered medical devices in operating rooms, transport vehicles, and outdoor medical settings
Smart Medical BMS
Real-time health monitoring, SOC/SOH reporting, and fault diagnostics — enabling smart medical batteries for connected healthcare equipment and battery management systems
Non-Toxic & RoHS Compliant
Environmentally responsible medical lithium battery chemistry — no heavy metals, no toxic electrolytes. The sustainable choice for medical batteries for sale in regulated markets
Medical Grade Standards
IEC 60601-1, IEC 62133, UN38.3, CE, UL, ISO 9001 — meeting medical device battery standards and medical device battery regulations for international healthcare markets
Medical Battery Technical Specifications
Key performance metrics for our LiFePO4 medical device batteries — from small battery-operated medical device packs to high-capacity medical equipment battery backup systems
Medical Battery Applications
Lithium battery solutions for the full range of medical equipment — from battery-powered medical devices in acute care to battery backup for home medical equipment and portable medical battery packs for field use
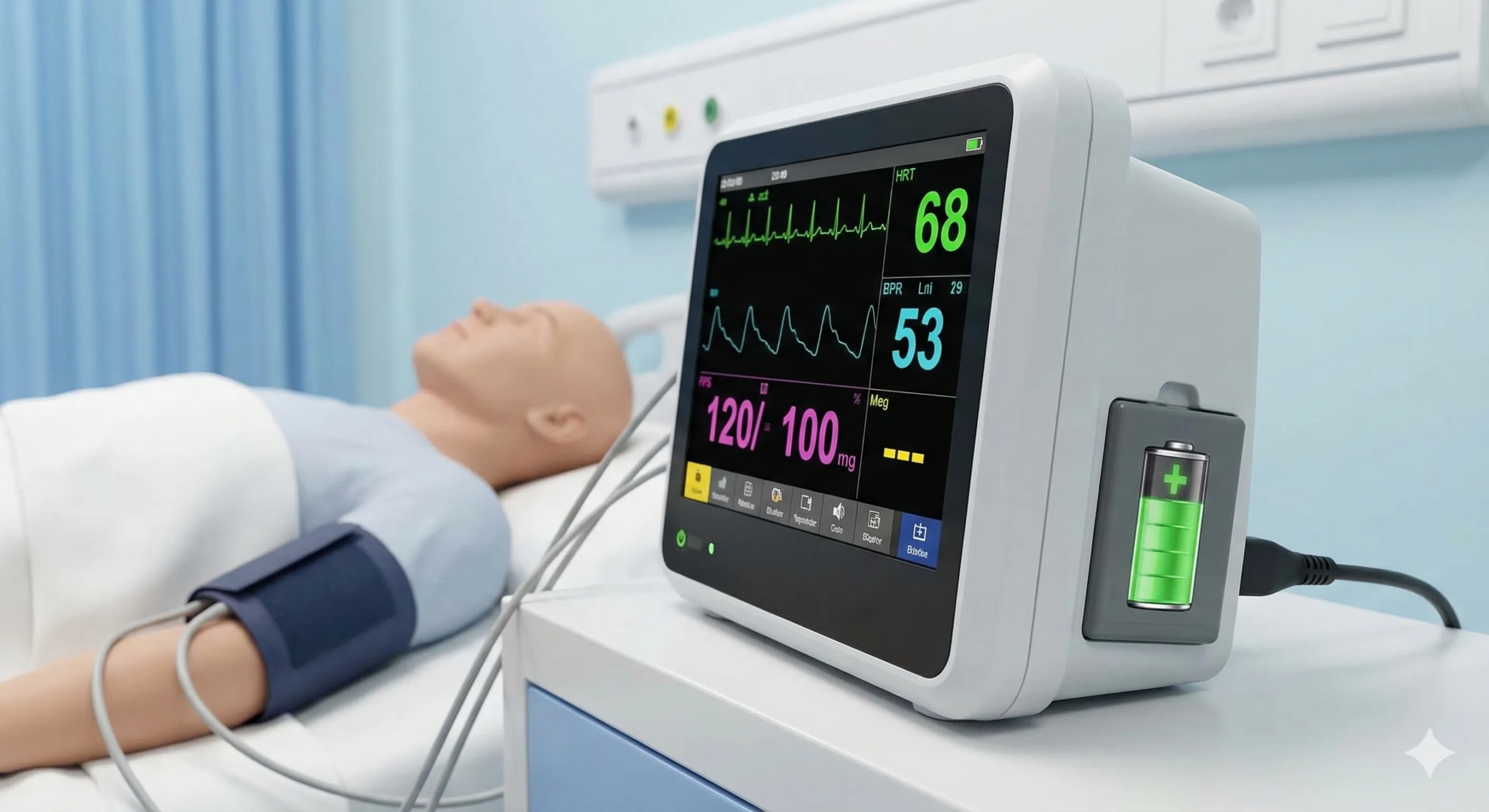
Patient Monitors
Reliable medical battery packs for vital signs monitors, ECG machines, SpO2 monitors, and patient tracking systems. GE Healthcare Marquette compatible medical battery options available. 12V–24V configurations with 4–8 hour runtime for ward and ICU use.
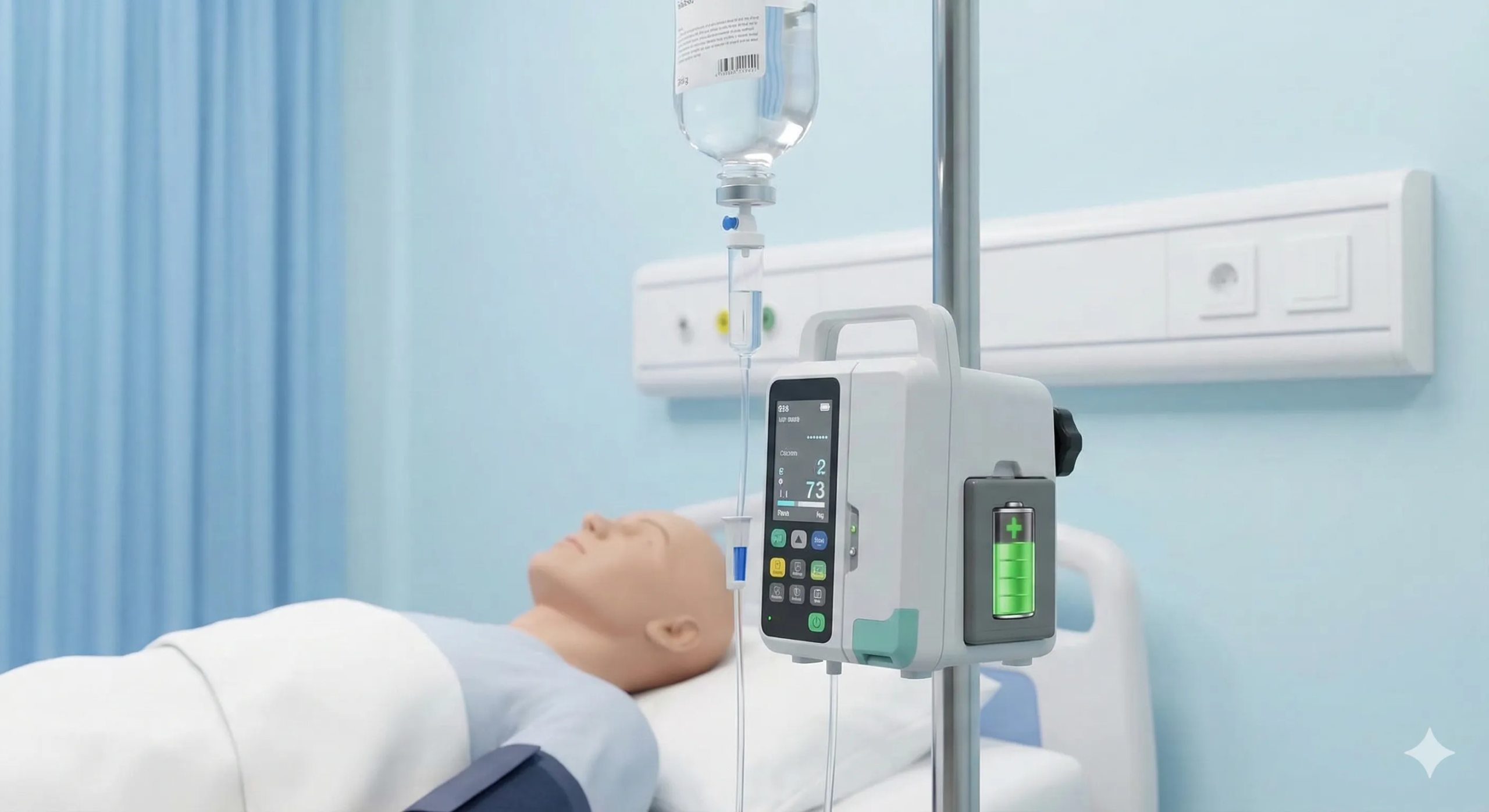
Infusion Pumps & Drug Delivery
Long-lasting lithium batteries for medication delivery systems, IV pumps, and syringe drivers. Precision medical battery design ensures stable power delivery — critical for accurate drug dosing in battery-powered medical devices used in hospital and home care settings.
Portable Ventilators
High-capacity battery-powered medical ventilator packs for respiratory support during transport and emergency situations. 24V–48V LiFePO4 configurations with 6–12 hour runtime — supporting battery backup systems for emergency medical equipment handling and field deployment.
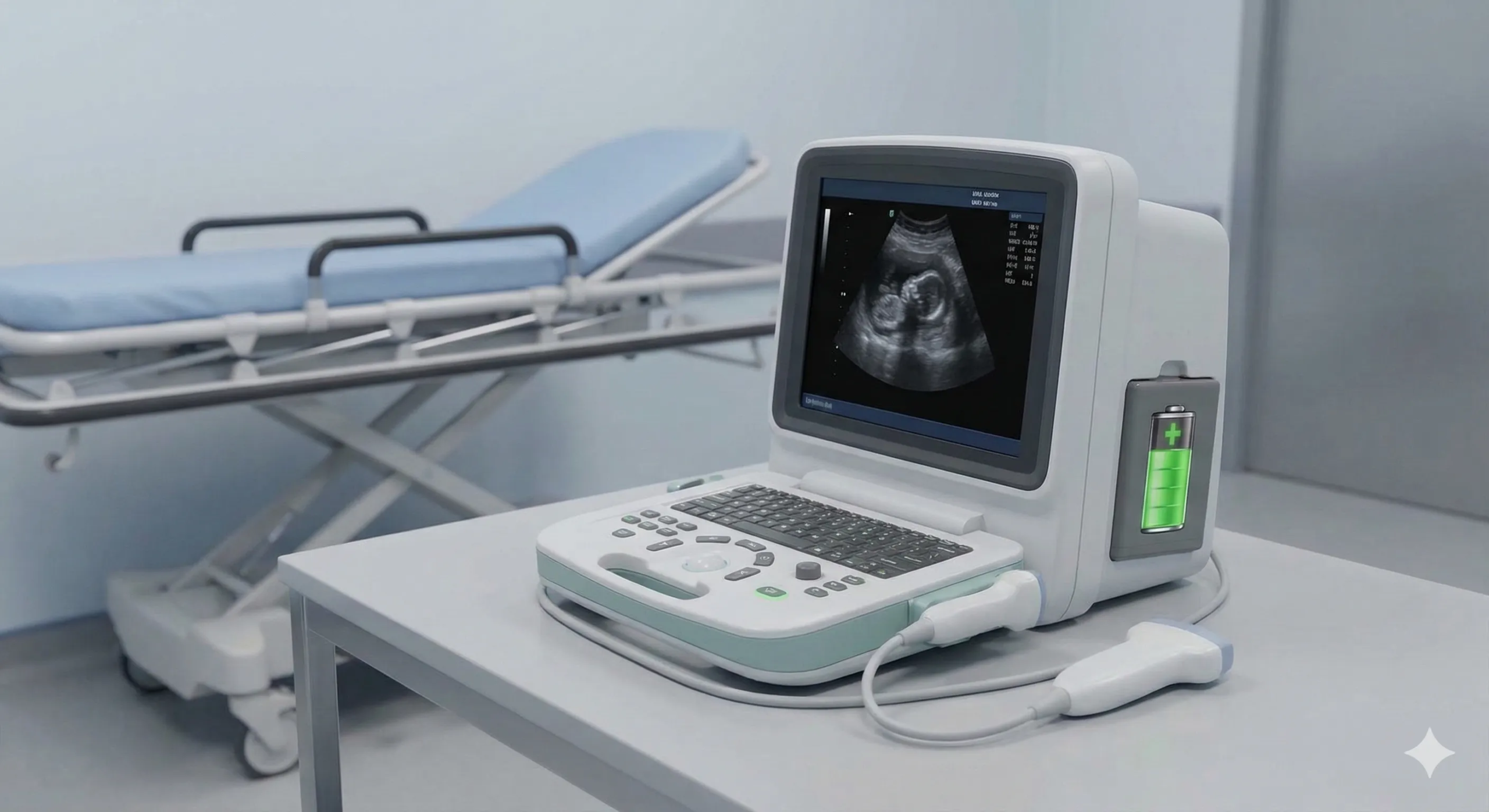
Diagnostic Equipment
Power solutions for portable ultrasound, portable X-ray, endoscopy equipment, and battery-powered medical optical devices. Custom lithium battery battery-powered medical optical device manufacturer — from design through certified production.

Mobility Aids & Medical Scooters
Medical scooter batteries, drive medical scooter battery, drive medical scout scooter battery, Roma medical mobility scooter battery, and e-wheels medical scooter battery. Replacement and OEM medical battery packs for power wheelchairs and mobility scooters.

Emergency & Critical Care Equipment
Reliable power for defibrillators, AEDs, battery-operated medical suction units, and emergency medical devices. Medical battery backup systems designed for zero-tolerance reliability when patient lives depend on uninterrupted power.
Quality Assurance, Cell Selection & Regulatory Compliance
Grade A Medical Cells — Fully Traceable
We use only Grade A LiFePO4 cells with medical-grade quality assurance and full lot traceability — supporting medical device battery requirements and regulatory compliance for medical battery manufacturers supplying hospitals, OEM device makers, and medical battery suppliers in international healthcare markets.
Comprehensive Medical Battery Testing
Every medical battery pack undergoes: capacity verification, internal resistance measurement, voltage matching, BMS protection function testing, thermal performance validation, and compliance testing against medical device battery standards. Full test reports available for all OEM and custom battery pack manufacturers for medical equipment.
Traceability & Lot Control in Medical Battery Manufacturing
Complete batch traceability from cell to finished medical battery pack — critical for regulatory submissions, post-market surveillance, and quality audits. ISO 9001-compliant production with full documentation for medical battery certification and international regulatory approval processes.

Certifications & Compliance for Medical Battery Markets








Medical Battery FAQ
This FAQ addresses questions about the legal and clinical concept of "medical battery" — the doctrine of unauthorized patient contact — which is frequently searched alongside questions about physical batteries used in medical devices. For product and technical questions about our medical device batteries, please contact our engineering team directly.
In a clinical or hospital setting, "medical battery" refers to the legal concept of non-consensual physical contact with a patient — for example, performing a medical procedure without the patient's informed consent. This is distinct from the physical batteries (power sources) used in medical devices and equipment. Healthcare institutions manage medical battery risk through robust consent protocols, documentation, and staff training. The medical definition of battery in healthcare law is intentional, harmful, or offensive touching without consent.
Medical battery involves unauthorized physical contact with a patient — performing a procedure without consent — regardless of the outcome. Medical negligence or malpractice involves harm caused by substandard care, even when consent was given. Battery claims can succeed even if the procedure was medically successful and caused no harm; negligence claims require proof of actual harm. Understanding the distinction between assault and battery in medical field settings is essential for healthcare risk management.
Informed consent is the primary defense against medical battery claims. When a patient provides valid, documented consent — understanding the nature, risks, benefits, and alternatives of a procedure — the provider is protected from battery allegations. Consent must be voluntary, competent, and specific to the procedure. General admission consent does not authorize specific invasive treatments. The battery definition in medical ethics centers on this consent requirement.
Any invasive procedure performed without specific patient consent carries medical battery risk — including surgery, anesthesia, blood transfusions, biopsies, catheterization, and certain examinations. Risk is highest when providers expand the scope of a procedure beyond what was consented to, or when treatment is administered to an incapacitated patient without following proper emergency or surrogate consent protocols. Medical battery examples in case law frequently involve surgical scope expansion.
Yes. Medical battery — in medical terms — is defined by the absence of consent, not by patient harm. A procedure performed without valid informed consent may constitute battery even if the patient experienced no adverse outcome and the treatment was medically appropriate. This is an example of medical battery that distinguishes it from malpractice, which requires proof of harm caused by a breach of the standard of care.
Strong documentation practices include: procedure-specific signed consent forms, detailed clinical notes of the consent discussion, notation of patient questions and responses, clearly defined scope of authorized treatment, documentation of patient decision-making capacity, and records of any authorized modifications to the treatment plan. Electronic health records with time-stamped entries provide the most defensible documentation in medical battery cases and medical battery malpractice litigation.
Hospitals and clinics should implement standardized consent protocols including: pre-procedure informed consent discussions in plain language; procedure-specific consent forms signed before treatment; verification of patient identity and decision-making capacity; documentation in the patient record; and a clear process for patients lacking capacity, including surrogate consent and advance directive review. Managing medical battery risk in medical ethics requires systematic, institutional-level consent governance — not just individual provider awareness.
Performing treatment beyond the scope of patient consent — for example, removing additional tissue during a consented procedure — can constitute medical battery and expose providers to civil liability, professional disciplinary action, and in some cases criminal charges. Courts apply a "reasonable patient" standard to assess whether the patient would have consented to the extended scope. Medical battery tort cases and medical battery lawsuits frequently arise from exactly this scenario. Medical battery statute of limitations varies by jurisdiction.
Effective training addresses: the legal and battery meaning in medical terms versus assault; proper informed consent procedures and documentation standards; recognizing treatment scope changes before proceeding; handling withdrawal of consent during a procedure; and escalation protocols when consent is unclear. Battery in medical ethics training should include case studies of medical assault and battery cases, simulation exercises, and regular compliance review. Battery on medical personnel and patient battery claims both carry serious institutional consequences.
Institutions must protect patients' rights to: refuse treatment at any time; receive clear information about proposed procedures; withdraw consent before or during a procedure; designate a healthcare proxy; have advance directives honored; and access their medical records. Systematic violations of these rights create institutional medical battery exposure and may generate medical battery cases that result in medical battery settlements. Medical battery consent is the cornerstone of these protections.
Yes. If a provider modifies a treatment plan in ways not covered by the original consent — particularly by adding interventions not discussed with the patient — this may constitute battery in the medical field. Providers should seek additional consent whenever treatment scope changes materially, even during surgery if the patient is under anesthesia, unless a genuine medical emergency makes this impossible. This is one of the most common medical battery examples in modern healthcare litigation.
Lack of consent — performing a procedure with no patient authorization — is the clearest form of medical battery in the medical field. Lack of informed consent involves obtaining agreement without adequately disclosing material risks, alternatives, or the nature of the procedure. The battery medical definition focuses on the former; cases involving inadequate disclosure are often litigated as negligence. Both carry serious legal risk and are addressed in medical battery law and medical battery court cases across jurisdictions.
When a patient is incapacitated and immediate treatment is necessary to prevent serious harm or death, providers may proceed under the "emergency exception" to consent. They should: attempt to contact the patient's surrogate or next of kin; consult available advance directives; document emergency circumstances and treatment rationale; and involve a second provider or ethics consultation where time permits. This exception is narrow and should not be applied to non-urgent situations — misapplication can still constitute medical assault and battery under medical law.
Effective organizational policies include: mandatory procedure-specific consent protocols; pre-surgical consent verification checklists; patient advocates or consent officers; standardized scope-of-treatment authorization language; escalation pathways when consent is disputed; regular audits of consent documentation; and defined processes for updating consent when treatment plans change. Risk management teams should systematically review medical battery vs assault incidents, near-misses, and medical battery in medical ethics education programs.
Contemporaneous, detailed records are the foundation of any medical battery defense. Key elements: signed procedure-specific consent forms; clinical notes documenting consent discussions and patient questions; records of capacity assessment; time-stamped electronic health record entries; nursing and anesthesia pre-operative consent verification; and written communication with patients or surrogates. Incomplete or altered records significantly weaken a provider's position in medical battery malpractice cases and medical battery court cases — and may themselves constitute grounds for disciplinary action.
